|
When two atomic 1 s orbitals combine in the formation of H 2, the result is two sigma ( σ) orbitals.Īccording to MO theory, one sigma orbital is lower in energy than either of the two isolated atomic 1 s orbitals –this lower sigma orbital is referred to as a bonding molecular orbital. Mathematical principles tell us that when orbitals combine, the number of orbitals before the combination takes place must equal the number of new orbitals that result from the combination – orbitals don’t just disappear! We saw this previously when we discussed hybrid orbitals: one s and three p orbitals make four sp 3 hybrids. A molecular orbital describes a region of space around two or more atoms inside which electrons are likely to be found. Recall that an atomic orbital (such as the 1s orbital of a hydrogen atom) describes a region of space around a single atom inside which electrons are likely to be found. In molecular orbital theory, we make a further statement: we say that the two atomic 1 s orbitals mathematically combine to form two new orbitals. When we described the hydrogen molecule using valence bond theory, we said that the two 1 s orbitals from each atom overlap, allowing the two electrons to be shared and thus forming a covalent bond. Let’s go back and consider again the simplest possible covalent bond: the one in molecular hydrogen (H 2). In order to understand these properties, we need to think about chemical bonding in a new way, using the ideas of molecular orbital (MO) theory. It fails to adequately account, for example, for some interesting properties of compounds that contain alternating double and single bonds. There are some areas, however, where the valence bond theory falls short. 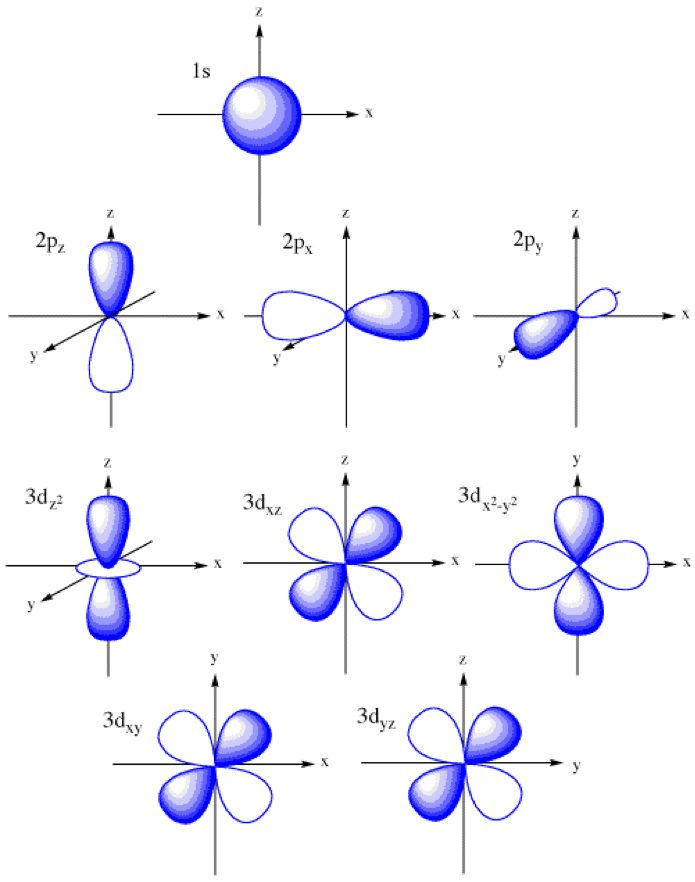
Valence bond theory does a remarkably good job at explaining the bonding geometry of many of the functional groups in organic compounds. 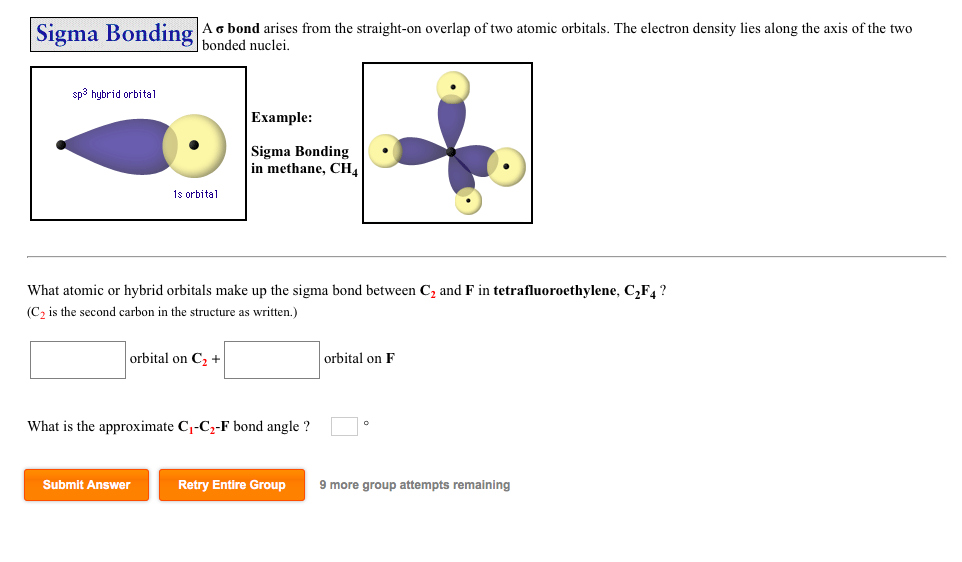
Molecular orbital theory, antibonding vs. This type of bond is referred to as a σ (sigma) bond. This means that the bond has cylindrical symmetry: if we were to take a cross-sectional plane of the bond at any point, it would form a circle. One more characteristic of the covalent bond in H 2 is important to consider at this point. The two overlapping 1 s orbitals can be visualized as two spherical balloons being pressed together. This ‘springy’ picture of covalent bonds will become very important in chapter 4, when we study the analytical technique known as infrared (IR) spectroscopy. It is not accurate, however, to picture covalent bonds as rigid sticks of unchanging length – rather, it is better to picture them as springs which have a defined length when relaxed, but which can be compressed, extended, and bent. 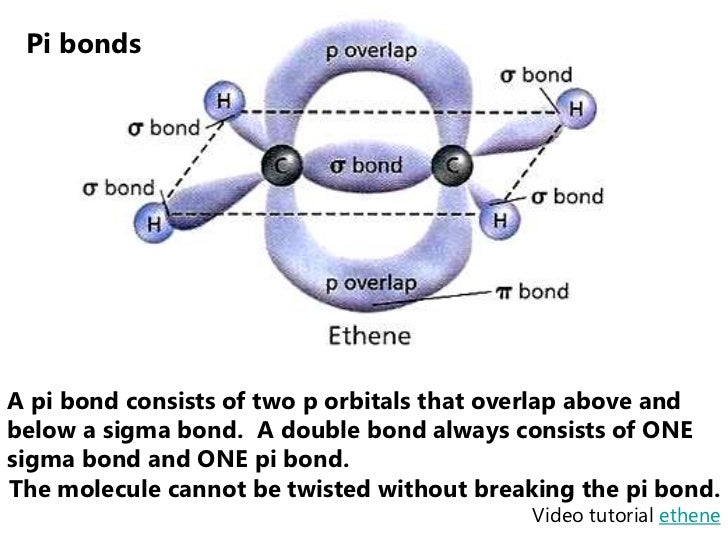
See a table of bond lengths and bond energies For the H 2 molecule, the distance is 74 pm (picometers, 10 -12 meters). Likewise, the difference in potential energy between the lowest energy state (at the optimal internuclear distance) and the state where the two atoms are completely separated is called the bond dissociation energy, or, more simply, bond strength. For the hydrogen molecule, the H-H bond strength is equal to about 435 kJ/mol.Įvery covalent bond in a given molecule has a characteristic length and strength. In general, the length of a typical carbon-carbon single bond in an organic molecule is about 150 pm, while carbon-carbon double bonds are about 130 pm, carbon-oxygen double bonds are about 120 pm, and carbon-hydrogen bonds are in the range of 100 to 110 pm. The strength of covalent bonds in organic molecules ranges from about 234 kJ/mol for a carbon-iodine bond (in thyroid hormone, for example), about 410 kJ/mole for a typical carbon-hydrogen bond, and up to over 800 kJ/mole for a carbon-carbon triple bond. There is a defined optimal distance between the nuclei in which the potential energy is at a minimum, meaning that the combined attractive and repulsive forces add up to the greatest overall attractive force. This optimal internuclear distance is the bond length. When the two nuclei are ‘too close’, we have an unstable, high-energy situation. \)Īt first this repulsion is more than offset by the attraction between nuclei and electrons, but at a certain point, as the nuclei get even closer, the repulsive forces begin to overcome the attractive forces, and the potential energy of the system rises quickly.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
